PRISMA Guidelines: Step-by-Step Workflow With Examples

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) is an evidence-based reporting guideline that helps researchers transparently document every phase of a systematic review. The PRISMA 2020 statement includes a 27-item checklist organized into seven sections and a four-phase flow diagram that tracks how records move from identification through screening to final inclusion. Following these guidelines ensures that readers, peer reviewers, and journal editors can evaluate exactly what was done, why it was done, and what was found. [1]
Systematic reviews hold a unique position in the evidence hierarchy because they synthesize findings across multiple studies rather than relying on a single investigation. However, the value of a systematic review depends entirely on how clearly it is reported. Incomplete reporting makes it impossible for others to assess the validity of the findings or replicate the process. PRISMA exists to solve this problem by providing a standardized framework that covers every aspect of reporting, from the rationale and search strategy to the risk of bias assessment and certainty of evidence. [2]
After reading this guide, you will understand what PRISMA requires, how to apply the 27-item checklist step by step, how to create a compliant flow diagram, and how to avoid the most common reporting mistakes in your own systematic review.
Key Takeaways

- PRISMA 2020 is a 27-item checklist and four-phase flow diagram designed to improve the transparency and completeness of systematic review reporting
- The checklist covers seven sections: title, abstract, introduction, methods, results, discussion, and other information
- The PRISMA flow diagram tracks how records move from identification through screening to inclusion, documenting exclusion reasons at each stage
- PRISMA is a reporting guideline, not a methodological tool. It tells you what to report, not how to conduct the review itself
- Extensions like PRISMA-S (for searches), PRISMA-ScR (for scoping reviews), and PRISMA-P (for protocols) address specific review types
What Are PRISMA Guidelines?

PRISMA is a reporting guideline, not a method for conducting a review. The distinction matters: PRISMA tells you what to include when you write up your systematic review, not how to design or execute one. [1]
The original PRISMA statement was published in 2009 and updated in 2020 to reflect advances in systematic review methodology over the previous decade. PRISMA 2020 replaces the earlier version and includes new items related to automation tools, risk of bias assessment, certainty of evidence, and protocol registration. [5]
The PRISMA 2020 statement consists of two main components. The first is a 27-item checklist that specifies what information should be reported in each section of a systematic review manuscript. The second is a flow diagram that visually maps how records move from the initial search through screening to final inclusion [1]. Most health science and social science journals now require or recommend PRISMA compliance as a condition for publication.
PRISMA is designed primarily for systematic reviews that evaluate the effects of interventions, but it can also be used as a foundation for reporting other types of reviews. Separate extensions provide tailored guidance for scoping reviews, network meta-analyses, diagnostic test accuracy reviews, individual participant data reviews, and protocol registrations. Researchers already familiar with AI-powered research tools for organizing evidence will find that PRISMA adds a structured reporting layer on top of any search and synthesis workflow.
PRISMA 2020 vs PRISMA 2009
The 2020 update introduced several significant changes to address gaps identified over the decade since the original statement. Understanding these differences is especially important if you are revising an older review or starting a new one.
| Feature | PRISMA 2009 | PRISMA 2020 |
|---|---|---|
| Checklist items | 27 items | 27 items with sub-items (more detailed) |
| Flow diagram versions | 1 template | 4 templates (new/updated, databases only/databases + other sources) |
| Protocol registration | Not explicitly required | Requires reporting registration info and deviations |
| Risk of bias | General mention | Requires reporting individual study and overall bias assessment methods |
| Certainty of evidence | Not included | Requires reporting certainty assessment (e.g., GRADE) |
| Automation tools | Not addressed | Requires reporting any automation used in study selection |
| Search reporting | Basic | Complemented by PRISMA-S extension with 16 items |
| Data availability | Not addressed | Requires reporting which data and code are available |
- The most notable additions in PRISMA 2020 are the requirements for reporting protocol deviations, certainty of evidence assessments, and the use of automation tools during screening. These changes reflect how systematic review practice has evolved, particularly with the growing role of machine learning in study identification and data extraction. [5]
The PRISMA 2020 Checklist (27 Items Explained)
The checklist is organized into seven sections. Each section corresponds to a part of your systematic review manuscript. Below is a summary of what each section requires.
Section 1: Title (Item 1)
The title must identify the report as a systematic review, meta-analysis, or both. Include the word "systematic review" explicitly so that databases and search engines index it correctly.
Section 2: Abstract (Item 2)
Provide a structured abstract that includes the objective, data sources, study eligibility criteria, participants, interventions, study appraisal methods, synthesis methods, results, limitations, conclusions, and registration information .
Section 3: Introduction (Items 3-4)
Describe the rationale for the review, referencing what is already known and what gaps exist. State the research questions and objectives, including the population, intervention, comparator, and outcome (PICO) framework where applicable.
Section 4: Methods (Items 5-16)
This is the largest section and requires the most detail. Report the following:
Eligibility criteria (Item 5). Define inclusion and exclusion criteria, including study design, participants, interventions, comparators, outcomes, and time frame.
Information sources (Item 6). List all databases, registers, websites, organizations, and reference lists searched, with the date of the last search for each. [4]
Search strategy (Item 7). Present the complete search strategy for at least one database, including any filters and limits applied.
Selection process (Item 8). Describe how many reviewers screened records, whether screening was independent, and what tools were used, including any automation.
Data collection (Item 9). Describe how data were extracted, how many reviewers extracted data, and how discrepancies were resolved.
Data items (Item 10). List all variables for which data were sought and any assumptions made about missing data.
Study risk of bias (Item 11). Report the tool used to assess risk of bias (e.g., RoB 2 for randomized trials, ROBINS-I for non-randomized studies) and how the assessment was applied.
Effect measures (Item 12). Specify the effect measures used, such as risk ratios, odds ratios, mean differences, or standardized mean differences.
Synthesis methods (Item 13). Describe how results were synthesized, whether through meta-analysis or narrative synthesis. AI-powered systematic review tools can assist with data extraction and synthesis, but you must report any such automation under this item. Report the statistical model, heterogeneity measures, and sensitivity analyses.
Reporting bias assessment (Item 14). Describe methods used to assess the risk of reporting bias across studies, such as funnel plots or the Egger test.
Certainty assessment (Item 15). Report the approach used to assess certainty of evidence, such as the GRADE framework.
Other methods (Item 16). Describe any additional methods not covered above, such as network meta-analysis or dose-response analysis.
Section 5: Results (Items 17-23)
Report the results of each stage.
Study selection (Item 17). Provide the number of records identified, screened, assessed for eligibility, and included, with reasons for exclusion. Present this information in the PRISMA flow diagram.
Study characteristics (Item 18). For each included study, report the citation, study design, setting, participants, intervention, comparator, outcomes, and funding source.
Risk of bias results (Item 19). Present risk of bias assessments for each included study and describe patterns across studies.
Individual study results (Item 20). Report summary data and effect estimates for each study, including confidence intervals.
Synthesis results (Item 21). Present the results of each synthesis, including meta-analysis forest plots, heterogeneity statistics, and narrative summaries where applicable.
Reporting bias results (Item 22). Present assessments of reporting bias for each synthesis.
Certainty of evidence (Item 23). Present the certainty of evidence for each outcome assessed. [2]
Section 6: Discussion (Items 24-26)
Provide a general interpretation of the results considering the body of evidence, any limitations of the evidence and the review process itself, and the implications for practice, policy, and future research.
Section 7: Other Information (Item 27)
Report protocol registration details, where the review protocol can be accessed, any deviations from the registered protocol, sources of funding, and the role of funders and sponsors.

How to Apply PRISMA (Step-by-Step Workflow)
Applying PRISMA is most effective when you plan for it from the beginning of your systematic review, not as an afterthought during manuscript writing. Follow this workflow.
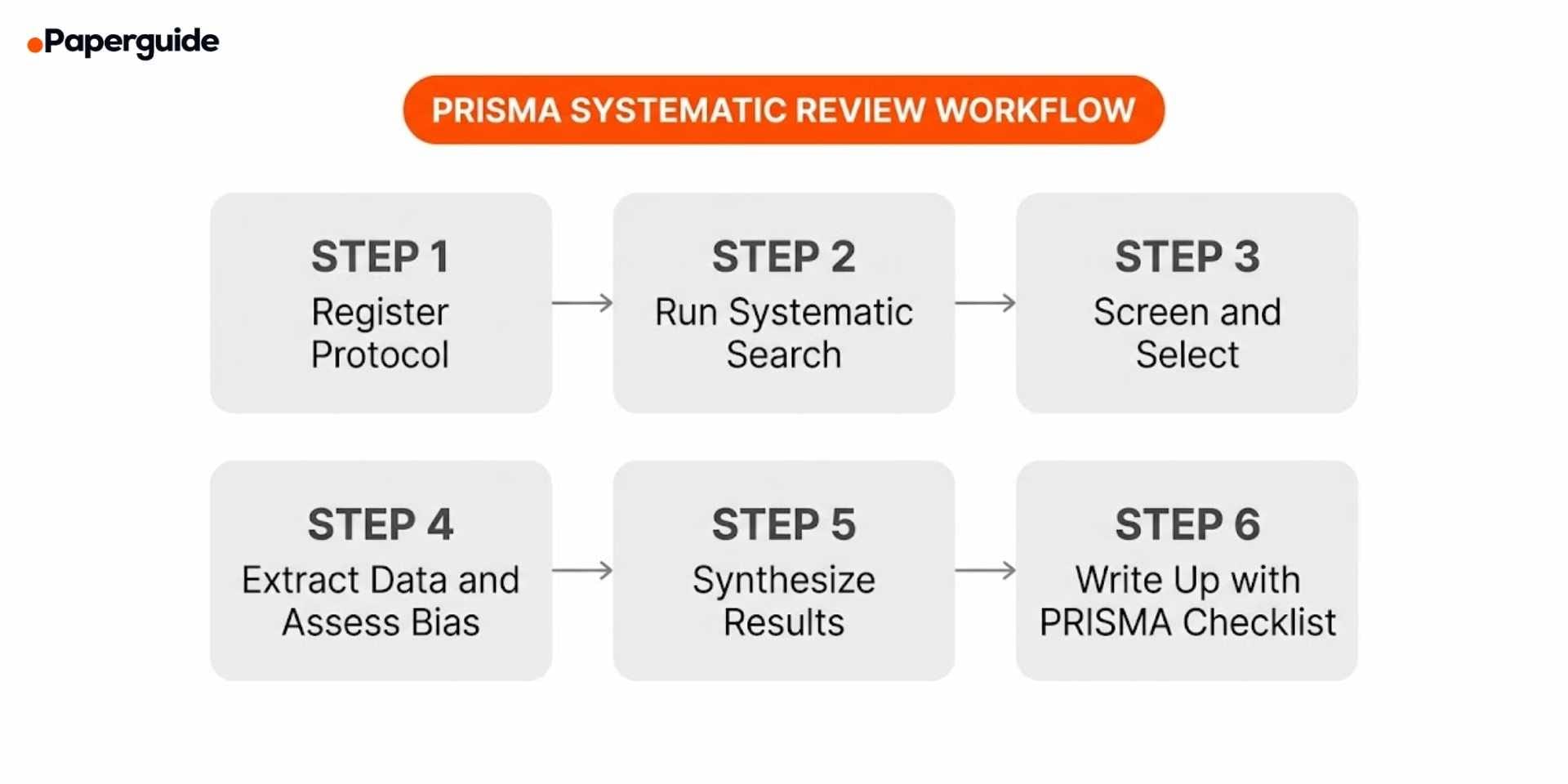
Step 1: Register Your Protocol
Before conducting your search, register the review protocol on PROSPERO (for health-related reviews) or another appropriate registry. The protocol should define your research question, eligibility criteria, search strategy, screening process, data extraction plan, and synthesis methods. PRISMA 2020 requires you to report any deviations from the registered protocol in the final manuscript [1].
Step 2: Run a Systematic Search
Search multiple databases and additional sources using a predefined strategy. Record the exact search terms, Boolean operators, filters, and date ranges for each database. The PRISMA-S extension provides 16 specific items for reporting your search strategy, including the platform and interface used, the full search string for each database, and any grey literature sources consulted. [4]
Keep a detailed log of how many records each source returned. This information feeds directly into the identification phase of the PRISMA flow diagram.
Step 3: Screen and Select Studies
Screen records in two stages. First, review titles and abstracts to exclude clearly irrelevant records. Second, obtain the full text of remaining records and assess them against your predefined eligibility criteria. Record the number of records excluded at each stage and the reasons for full-text exclusions.
If you use automation tools such as machine learning classifiers to assist with screening, PRISMA 2020 requires you to report the tool, how it was used, and how its recommendations were verified. Platforms that combine reference management with screening workflows help keep these numbers organized from the start. Researchers managing large reference libraries during screening often benefit from tools that integrate AI-powered search with reference management to maintain accurate counts at every stage. [5]
Step 4: Extract Data and Assess Risk of Bias
Extract data from each included study using a standardized form. At a minimum, extract study design, setting, participants, interventions, comparators, outcomes, and results. Record who extracted the data, whether extraction was done independently by multiple reviewers, and how disagreements were resolved.
Assess risk of bias for each included study using a validated tool. For randomized trials, RoB 2 is the current standard. For non-randomized studies, ROBINS-I is recommended. Report the risk of bias results both for individual studies and as patterns across the entire body of evidence. For a deeper comparison of how different platforms support evidence synthesis and consensus-based research, consider how your chosen tools handle bias documentation at scale. [2]
Step 5: Synthesize Results
Synthesize the extracted data either quantitatively through meta-analysis or narratively through structured summary. If performing a meta-analysis, report the statistical model (fixed or random effects), the measure of heterogeneity (I-squared, tau-squared), and any subgroup or sensitivity analyses. If a meta-analysis is not appropriate, use a structured narrative synthesis and explain why quantitative pooling was not feasible.
Assess reporting bias across studies using methods such as funnel plots or statistical tests. Report the certainty of evidence for each outcome using a framework like GRADE.
Step 6: Write Up Your Review Using the PRISMA Checklist
Go through the 27-item checklist systematically as you draft your manuscript. For each item, verify that you have reported the required information and note the page number or section where it appears. Many journals now require a completed PRISMA checklist as a supplementary file at submission. [3]
The PRISMA Flow Diagram (With Example)
The PRISMA flow diagram is a visual summary of the study selection process. It tracks records through four phases: identification, screening, eligibility, and inclusion.
PRISMA 2020 provides four flow diagram templates depending on whether the review is new or updated and whether searches included databases only or databases plus other sources. For most new reviews using databases and registers, use the standard template from the PRISMA website. [1]
Example PRISMA Flow Diagram (Completed)
Below is a filled example for a systematic review on mindfulness interventions for academic stress.
Identification Records identified from databases (n = 2,847): PubMed (n = 892), PsycINFO (n = 1,104), Scopus (n = 851) Records identified from registers (n = 43): ClinicalTrials.gov (n = 43)
Before screening Records removed before screening: Duplicates (n = 687), Marked as ineligible by automation tools (n = 0), Other reasons (n = 0)
Screening Records screened by title and abstract (n = 2,203) Records excluded (n = 1,891)
Retrieval Reports sought for retrieval (n = 312) Reports not retrieved (n = 14)
Eligibility Reports assessed for full-text eligibility (n = 298) Reports excluded with reasons (n = 264): Wrong population (n = 89), Wrong intervention (n = 71), Wrong outcome (n = 52), Wrong study design (n = 34), Duplicate data (n = 18)
Included Studies included in the review (n = 34) Studies included in meta-analysis (n = 28)
This diagram gives readers a complete picture of how you moved from thousands of initial records to the final included studies, with documented reasons for every exclusion.
PRISMA Reporting Template
Use this fill-in template to ensure your systematic review covers each required PRISMA section. Replace the bracketed sections with your own content.
Title: [Systematic review/meta-analysis] of [intervention or exposure] for [population or condition]: a systematic review [and meta-analysis].
Registration: This review was registered on [registry name] with registration number [number]. The protocol is accessible at [URL or DOI].
Search Strategy: We searched [list databases] from [start date] to [end date] using the following terms: [list key terms and Boolean operators]. The complete search strategy for [primary database] is provided in Supplement [number].
Eligibility Criteria: We included [study designs] that examined [intervention/exposure] in [population] and reported [outcomes]. We excluded [list exclusion criteria].
Screening Process: Two reviewers independently screened [number] titles and abstracts, followed by full-text review of [number] reports. Disagreements were resolved by [method].
Risk of Bias: We assessed risk of bias using [tool name]. Each study was rated as [low/some concerns/high] for each domain. Results are presented in [figure/table number].
Synthesis: We conducted [meta-analysis using random/fixed effects model / narrative synthesis]. Heterogeneity was assessed using [I-squared and tau-squared]. Subgroup analyses were performed for [variables]. Certainty of evidence was assessed using [GRADE or equivalent].
Flow Diagram: The PRISMA flow diagram (Figure [number]) shows that [total identified] records were identified, [total screened] were screened, and [total included] studies were included.
Filled Example:
Title: Effectiveness of mindfulness-based interventions for reducing academic stress in university students: a systematic review and meta-analysis.
Registration: This review was registered on PROSPERO with registration number CRD42024000123. The protocol is accessible at https://www.crd.york.ac.uk/prospero.
Search Strategy: We searched PubMed, PsycINFO, and Scopus from January 2015 to December 2025 using the following terms: (mindfulness OR meditation OR MBSR) AND (academic stress OR exam anxiety OR student wellbeing). The complete search strategy for PubMed is provided in Supplement 1.
Eligibility Criteria: We included randomized controlled trials and quasi-experimental studies that examined mindfulness-based interventions in undergraduate or graduate students and reported stress or anxiety outcomes. We excluded case reports, qualitative studies, and interventions shorter than four weeks.
Screening Process: Two reviewers independently screened 2,203 titles and abstracts, followed by full-text review of 298 reports. Disagreements were resolved by discussion and, when necessary, consultation with a third reviewer.
Risk of Bias: We assessed risk of bias using RoB 2. Each study was rated as low risk, some concerns, or high risk for each domain. Results are presented in Figure 3.
Synthesis: We conducted a meta-analysis using a random effects model. Heterogeneity was assessed using I-squared and tau-squared. Subgroup analyses were performed for intervention duration and student level. Certainty of evidence was assessed using the GRADE framework.
Flow Diagram: The PRISMA flow diagram (Figure 1) shows that 2,890 records were identified, 2,203 were screened, and 34 studies were included
Common PRISMA Mistakes

Mistake 1: Incomplete or Incorrect Flow Diagram
The flow diagram is the most visible part of PRISMA compliance, yet many reviews submit diagrams with numbers that do not add up. The total records minus duplicates should equal the number screened. The number screened minus exclusions should equal the number retrieved. Each subtraction must be traceable.
Fix: Build your flow diagram alongside the review process, updating it after each phase. Double-check that all numbers reconcile before submission. Use the official PRISMA 2020 templates from prisma-statement.org.
Mistake 2: Not Registering the Protocol
PRISMA 2020 requires authors to report whether and where the protocol was registered. Reviewers who skip protocol registration cannot demonstrate that their methods were predefined, which raises concerns about selective reporting and post-hoc changes to eligibility criteria. [3]
Fix: Register your protocol on PROSPERO or a similar platform before beginning your search. Include the registration number and a link to the protocol in your manuscript.
Mistake 3: Reporting Databases Without the Full Search Strategy
Stating that you "searched PubMed, Scopus, and Web of Science" without providing the actual search strings makes it impossible for anyone to replicate or evaluate your search. PRISMA 2020 requires the full search strategy for at least one database, and the PRISMA-S extension recommends providing it for all databases. [4]
Fix: Include the complete search string with Boolean operators, MeSH terms, truncation, and filters for at least one database in the methods section. Provide strategies for additional databases in a supplementary file.
Mistake 4: Skipping Risk of Bias Assessment
Some reviews report results without assessing the risk of bias in the included studies. Without this assessment, readers cannot judge whether the findings are trustworthy or driven by studies with serious methodological limitations.
Fix: Use a validated risk of bias tool (RoB 2 for randomized trials, ROBINS-I for non-randomized studies). Present results for each study and summarize patterns across the body of evidence.
Mistake 5: Treating PRISMA as a Methodology Guide
PRISMA is a reporting guideline. It does not tell you how to formulate your research question, which databases to search, or how to conduct a meta-analysis. Researchers who use PRISMA as a substitute for methodological guidance (such as the Cochrane Handbook) often produce reviews with sound reporting but weak methods.
Fix: Use established methodological guidance (Cochrane Handbook, JBI Manual, or GRADE handbook) to design and conduct your review. Use PRISMA to report what you did and what you found. [3]
PRISMA Quality Checklist
- [ ] Title identifies the review type. The title explicitly states that the manuscript is a systematic review, meta-analysis, or both.
- [ ] Abstract is structured. The abstract follows a structured format covering objectives, methods, results, and conclusions.
- [ ] Protocol is registered. The registration number, registry name, and protocol access information are provided.
- [ ] Search is fully reported. The complete search strategy for at least one database is included, with dates and all terms.
- [ ] Screening process is documented. The number of reviewers, independence of screening, and conflict resolution methods are described.
- [ ] Risk of bias is assessed. A validated tool was used and results are reported for each included study.
- [ ] Synthesis methods are specified. The statistical model, heterogeneity measures, and sensitivity analyses are reported.
- [ ] Certainty of evidence is assessed. The GRADE framework or equivalent was applied and results are presented per outcome.
- [ ] Flow diagram is complete. Numbers reconcile across all phases, and exclusion reasons with counts are provided at full-text stage.
- [ ] Protocol deviations are documented. Any differences between the registered protocol and the final review are explained.
PRISMA Extensions for Different Review Types
PRISMA 2020 primarily covers systematic reviews of interventions, but extensions provide guidance for other review types and aspects.
| Extension | Purpose | Key Addition |
|---|---|---|
| PRISMA-S | Search reporting | 16 items for documenting literature searches |
| PRISMA-P | Protocols | Checklist for systematic review protocols |
| PRISMA-ScR | Scoping reviews | Adapted items for scoping review reporting |
| PRISMA-DTA | Diagnostic test accuracy | Items for reporting diagnostic accuracy reviews |
| PRISMA-IPD | Individual participant data | Guidance for IPD meta-analyses |
| PRISMA-NMA | Network meta-analyses | Items for reporting network comparisons |
| PRISMA-Harms | Adverse effects | Checklist for reporting harms in systematic reviews |
| PRISMA-Equity | Health equity | Items for reviews focused on equity considerations |
Choose the extension that matches your review type and use it alongside the main PRISMA 2020 checklist. For literature reviews that do not follow the full systematic review methodology, refer to alternative academic writing and review tools that support less structured review formats.
Validate This With Papers (2 Minutes)
Before submitting your systematic review manuscript, verify your PRISMA compliance against published examples and the official checklist to catch any reporting gaps.
Step 1: Download the official PRISMA 2020 checklist from prisma-statement.org. Go through each of the 27 items and confirm that your manuscript addresses it. Note the page number or section for each item, as many journals require this as a supplementary table.
Step 2: Search for two or three recently published PRISMA-compliant systematic reviews in your field. Compare their reporting structure against yours. Pay attention to how they document their search strategy, present risk of bias tables, and structure their flow diagrams. Paperguide's AI Academic Search can help you quickly locate published systematic reviews on your topic and pull relevant methodological details from across multiple papers.
Step 3: For specific items you are unsure about, use Scholar GPT to search for the PRISMA 2020 explanation and elaboration paper and ask targeted questions such as "What should Item 13a include?" or "How should I report deviations from the protocol?" This gives you direct answers grounded in the official PRISMA guidance without reading the entire 80-page document.
This takes about two minutes and significantly reduces the risk of a desk rejection for incomplete PRISMA reporting.
Conclusion
PRISMA 2020 provides the definitive framework for reporting systematic reviews and meta-analyses transparently and completely. Its 27-item checklist and four-phase flow diagram ensure that every aspect of the review process, from the initial search strategy through screening, data extraction, risk of bias assessment, synthesis, and certainty of evidence, is documented in a way that allows readers to evaluate and replicate the work. The guidelines do not dictate how to conduct a review, but they set the standard for how to communicate what was done. For any researcher conducting a systematic review or meta-analysis, understanding PRISMA is not optional. It is the baseline expectation of every major journal.
If you are starting your first systematic review, begin with protocol registration, plan your search strategy with PRISMA-S in mind, and keep the checklist beside you from the first draft through final submission. Whether you are working on a thesis, a journal manuscript, or a grant-funded review, the process is the same. The difference between a review that gets desk-rejected and one that moves to peer review often comes down to the completeness of PRISMA reporting. The checklist, flow diagram templates, and extensions described in this guide give you everything needed to meet that standard from day one.
Frequently Asked Questions
Is PRISMA required for all systematic reviews?
PRISMA is not legally required, but it is the standard expected by most journals in health sciences, social sciences, and education. Many journals explicitly require a completed PRISMA checklist at submission. Even when not mandatory, following PRISMA significantly improves the quality and transparency of your manuscript, which increases the likelihood of acceptance [3].
What is the difference between PRISMA and the Cochrane Handbook?
PRISMA is a reporting guideline that tells you what to include in your manuscript. The Cochrane Handbook is a methodological guide that tells you how to conduct a systematic review. You need both. Use the Cochrane Handbook (or equivalent guidance like the JBI Manual) to design and conduct the review, then use PRISMA to report it.
Can I use PRISMA for a narrative literature review?
PRISMA is designed for systematic reviews and meta-analyses, not narrative literature reviews. However, narrative reviewers can adapt elements of PRISMA, particularly the flow diagram and search documentation, to improve transparency. For scoping reviews, use the PRISMA-ScR extension instead of the main PRISMA 2020 statement.
When should I start using the PRISMA checklist?
Start at the protocol stage. While PRISMA is technically a reporting tool, planning your review with the checklist in mind ensures you collect and document all the information you will need during write-up. Researchers who leave PRISMA compliance to the end often discover they failed to record critical details during earlier stages.
How do I complete the PRISMA flow diagram?
Download the appropriate template from prisma-statement.org. There are four versions depending on whether your review is new or updated and whether you searched databases only or databases plus other sources. Fill in the numbers as you progress through each screening phase. Verify that all numbers add up correctly before submission.
What is PRISMA-S and do I need it?
PRISMA-S is an extension to the PRISMA statement specifically for reporting literature searches. It includes 16 items covering the database name and platform, the full search strategy, any limits applied, the date of each search, and the management of search results. If your review includes a systematic literature search, following PRISMA-S alongside PRISMA 2020 strengthens the reproducibility of your search methodology.
Does PRISMA apply to scoping reviews?
Not directly. The main PRISMA 2020 statement is designed for systematic reviews of interventions. For scoping reviews, use the PRISMA-ScR (PRISMA Extension for Scoping Reviews) checklist, which was published in 2018 and adapts PRISMA items to the scoping review methodology. The two checklists share a similar structure but differ in items related to risk of bias and evidence synthesis, which are typically not part of scoping reviews.
References
- Page, M.J., McKenzie, J.E., Bossuyt, P.M. et al. "The PRISMA 2020 statement: an updated guideline for reporting systematic reviews." BMJ, 2021.
- Page, M.J., Moher, D., Bossuyt, P.M. et al. "PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews." BMJ, 372, 2021.
- Sarkis-Onofre, R., Catalá-López, F., Aromataris, E. & Lockwood, C. "How to properly use the PRISMA Statement." Systematic Reviews, 10(1), 2021.
- Rethlefsen, M.L., Kirtley, S., Waffenschmidt, S. et al. "PRISMA-S: an extension to the PRISMA statement for reporting literature searches in systematic reviews." Systematic Reviews, 10(1), 2021.
- Page, M.J., McKenzie, J.E., Bossuyt, P.M. et al. "Updating guidance for reporting systematic reviews: development of the PRISMA 2020 statement." Journal of Clinical Epidemiology, 134, 2021.





